Steroid hormones control the process of growth and development and other physiological response in different types of cells and tissues. This control process of steroid hormone follows the steroid hormone receptor attraction mechanism, which activates the different types of genes at transcription and post-transcription process (Lodish et al., 2000).
There are almost 48 steroid hormone and nuclear receptors (NRs) are found in the humans. The most commonly known steroid hormone receptors are estrogen receptor (ERα and ERβ), glucocorticoid receptor (GR), mineralocorticoid receptor, progesterone receptor (PR) and androgen receptor (AR). Cholesterol is the precursor of all these steroid hormones. All steroid hormones are mainly secreted by the endocrine gland like adrenal cortex and from gonads. After they secreted from the glands, they are diffused into the bloodstream. Lipid soluble steroid hormones easily pass through the cellular membrane and can bind to the steroid hormone receptors which are located in their target area and play a wide range of functions like cell homeostasis, differentiation and regulation of proliferation, survival and cell death (Evans & Mangelsdorf, 2014).
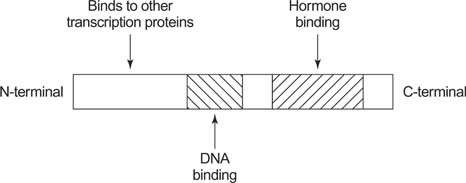
Steroid receptors are present at different locations in the target cell. Progesterone an estrogen receptors are mainly present in the target cell nucleus. On the other hand, glucocorticoid receptor and androgen receptor are located in the target cell cytoplasm Steroid receptors possess structural similarity and their way of an act as a transcriptional factor to the nuclear receptor superfamily as they belong to this family. The steroid hormone receptor consists of four domains. These domains are a variable N-terminal domain, a DNA binding domain, a hinge region and a hormone/ligand binding domain. Nuclear/cytoplasmic movement of these receptors is carried out with the help of nuclear localization signals (NLS) within the hinge region (Ward & Weigel, 2009).
The mechanism of action of steroid hormone receptors is summarized as follow:
First, free lipid soluble steroid hormone passes through the target cells plasma membrane. Then it binds to the steroid hormone receptor which mainly present in the cytoplasm or nucleus. Steroid hormone receptor contains hormone binding domain and DNA binding domain. When steroid hormone binds to its receptor, then this hormone-receptor complex bind to the specific region of DNA called Hormone response element (HRE) with the help of DNA binding domain of the receptor molecule. After the attachment to these specific sites of DNA, they trigger or stop the mRNA formation from these genes. Then new mRNA move out from the nucleus to the cytoplasm. Translation of mRNA occurs and new desired protein is formed which ultimately show an effective response (Zheng & Murphy, 2016).

Moreover, steroid hormones stimulate the many intracellular signalling pathways when they bind to receptors associated with the membrane. This is a non-genomic action of steroid hormones. In addition, transcription of many target genes also followed the ligand-independent pathway by the steroid receptors. In this mechanism, steroid receptors and their co-regulators are phosphorylated by the active protein kinases. Steroid receptors enhance or suppress the transcription of the gene by the presence of coactivators or corepressors which are co-regulators. Commonly known co-regulators are steroid receptor coactivator (SRC) family, steroid receptor RNA activator (SRA), androgen receptor-associated proteins (ARAs) and the PIAS (protein inhibitor of an activated signal transducer and activator of transcription) family (Whirledge & Cidlowski, 2017).
When the hormone receptors are phosphorylated, they are active and activate the mechanism of cell signalling pathways. When they are active, they regulate the stability of the protein, localization in the nucleus, sensitivities of hormones, binding to DNA and transcriptional process (Weigel & Moore, 2007). In all domains of steroid receptors phosphorylation occur, but the majority of phosphorylation was reported in the N-terminal domain of steroid hormone receptor (Ward & Weigel, 2009).

In the process of long-term response to stress, an adrenocorticotropic hormone is released from the anterior pituitary gland. These hormones trigger the release of corticosteroid hormones from the adrenal cortex. In this response, transcription of many genes starts in the target cell nuclei and results in the changes of concentration of enzymes in the cytoplasm and helps in cellular metabolism (Van der Velden, 1998).
Another steroid hormone, glucocorticoid binds to the glucocorticoid receptors. This hormone-receptor complex produced anti-inflammatory action when binds to a promoter region of glucocorticoid-responsive elements or by the interaction with transcription factors. This complex activates the gene protein-1 or nuclear factor-kappa B. This then blocked the action of inflammatory molecules like cytokines, chemokines, arachidonic acid metabolites, and adhesion molecules (Van der Velden, 1998).
References
Evans, R. M., & Mangelsdorf, D. J. (2014). Nuclear receptors, RXR, and the big bang. Cell, 157 (1), 255-266.
Lodish, H., Berk, A., Zipursky, S., Matsudaira, P., Baltimore, D., & Darnell, J. (2000). Overview of extracellular signaling. Molecular Cell Biology, 4.
Ward, R. D., & Weigel, N. L. (2009). Steroid receptor phosphorylation: Assigning function to site‐specific phosphorylation. Biofactors, 35 (6), 528-536.
Weigel, N. L., & Moore, N. L. (2007). Kinases and protein phosphorylation as regulators of steroid hormone action. Nuclear receptor signaling, 5.
Whirledge, S., & Cidlowski, J. A. (2017). Steroid Hormone Receptors Act as Ligand-Dependent Transcription Factors or Repressors. Yen & Jaffe’s Reproductive Endocrinology E-Book: Physiology, Pathophysiology, and Clinical Management, 115.
Van der Velden, V. (1998). Glucocorticoids: mechanisms of action and anti-inflammatory potential in asthma. Mediators of inflammation, 7 (4), 229-237.
Zheng, Y., & Murphy, L. C. (2016). Regulation of steroid hormone receptors and coregulators during the cell cycle highlights potential novel function in addition to roles as transcription factors. Nuclear receptor signaling, 14.
















